 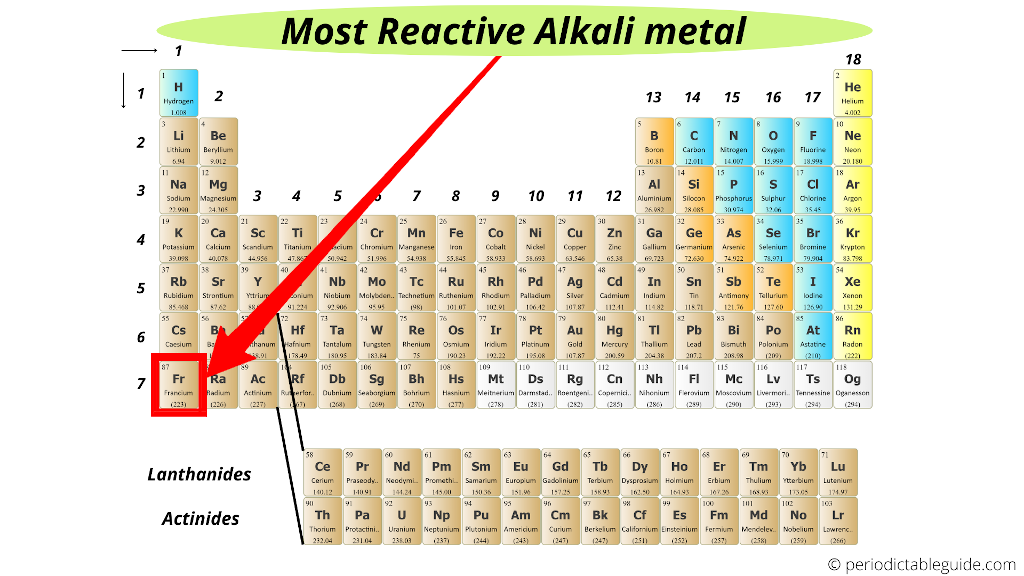
For example, an element lower down in group 1, like caesium, will be more reactive than an element higher up in group one, like potassium. There are different trends in reactivity for metals and non-metals and often these trends are opposite. This is hard to answer because it depends on whether we are discussing a non-metal or metal. The most reactive group 7 element is Fluorine. This is because the electron is attracted by the positive nucleus so the shorter the distance to the nucleus, the better. However, in this 'gaining electron' scenario, the most reactive element in group 7 is the one with the fewest electron shells. Similarly, the elements that find it easiest to gain an electron are those with 7 electrons in their outer shell (all elements in group 7 as they really want to have 8 electrons in their outer shell). Therefore, all group 1 elements are the very reactive (they all have 1 electron in their outer shell) but Francium is the most reactive (it has the most number of electron shells therefore there is a greater distance between the outer electron and the nucleus). It is easiest for an atom to lose an electron (therefore easier for it to react/be more reactive) if there is only one electron in the outer shell and there is a big distance from the nucleus. 
Ionic reactions tend to be more vigorous than covalent. A chemical reaction always includes a reshuffle of electrons (which are found in orbiting the positive protons and neutrons in the nucleus). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
